-
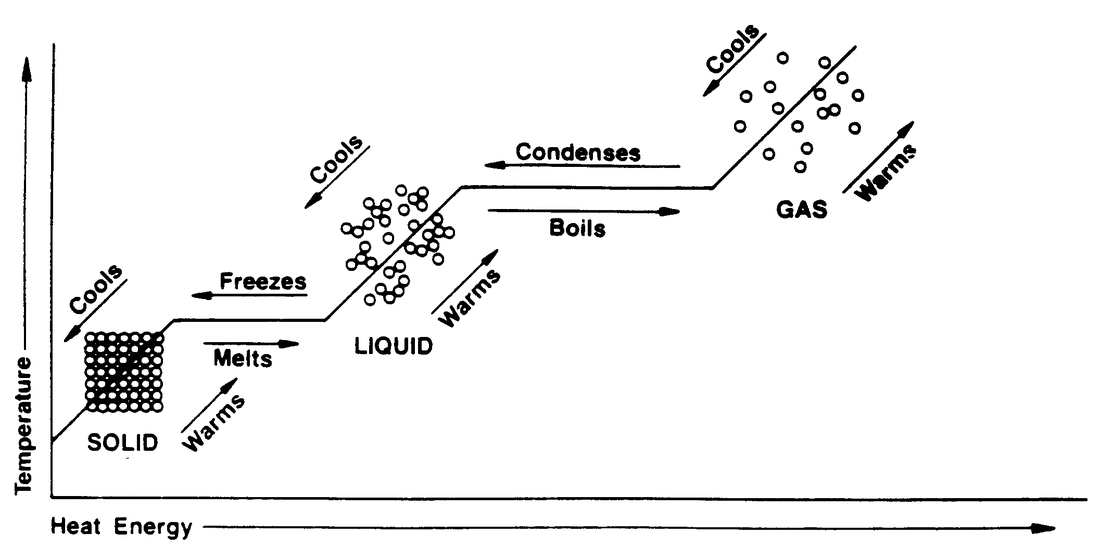
Melting/Freezing/Boiling/Condensation
-
Density
-
Mass and Volume
-
Other Properties
<
>
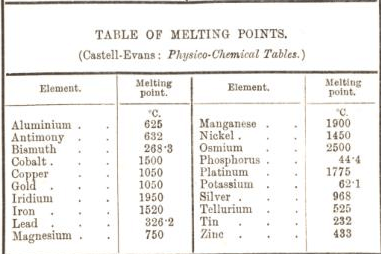
Boiling Point and Melting Point
|
Which substances have...
...a very high melting point? ...a very low melting point? |
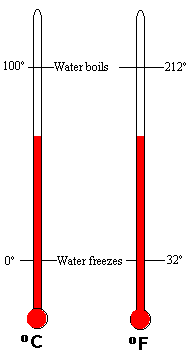
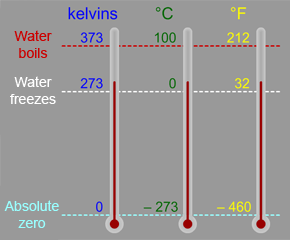
With this video, it helps to know what the Kelvin temperature scale is.
|
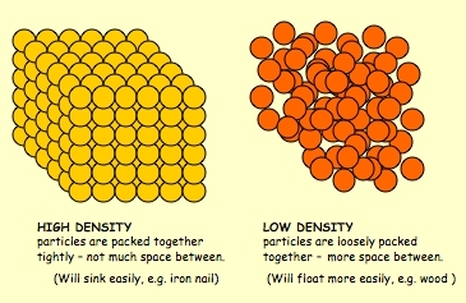
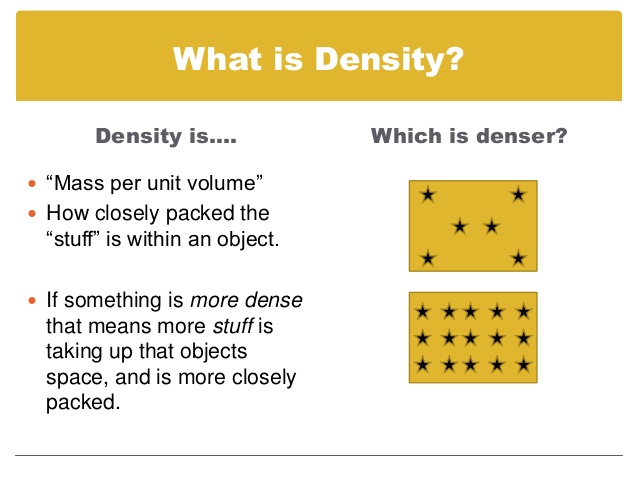
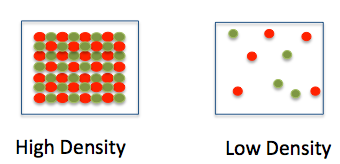
Density
|
How would you explain DENSITY to someone? Try doing it again, only use the words MASS and VOLUME. Now try doing it WITHOUT using those words. Can you? |
|
|
"...but steel is heavier than feathers!"
|
Limmy is having trouble with this misconception. He keeps saying, "But steel is heavier than feathers."
What he should be thinking is, "Steel is more dense than feathers." Someone explain it to him already! |
|
How many different densities can we layer?
How do the particles in honey compare to the particles of oil? |
Some sink, some float...
........why did this happen? |
|
|
|
|
Interesting questions: Do you think it is possible for two things to have the EXACT SAME density? If they did, would either one try to float or sink? So how does this freaky drink from the 90s actually work? |
|
THE CROWN OF SYRACUSE: How Density Solved a Mystery
|
|
In ancient Greece,
a king wanted a crown of pure gold. How does he know if it's pure? How can knowing the MASS, VOLUME, and DENSITY help solve this mystery? |
|
Great density demonstration! KEY QUESTION: What is the relationship between mass and volume?
MASS
WEIGHT
|
Sometimes, we use weight to measure mass, but they are not the same!
Weight measures how hard gravity pulls on an object. So on Earth, I may weigh a certain amount, but if I go somewhere with different gravity, my weight can change! Notice how no matter where you go, mass stays the same. That's because you're still made of the same atoms and molecules! |
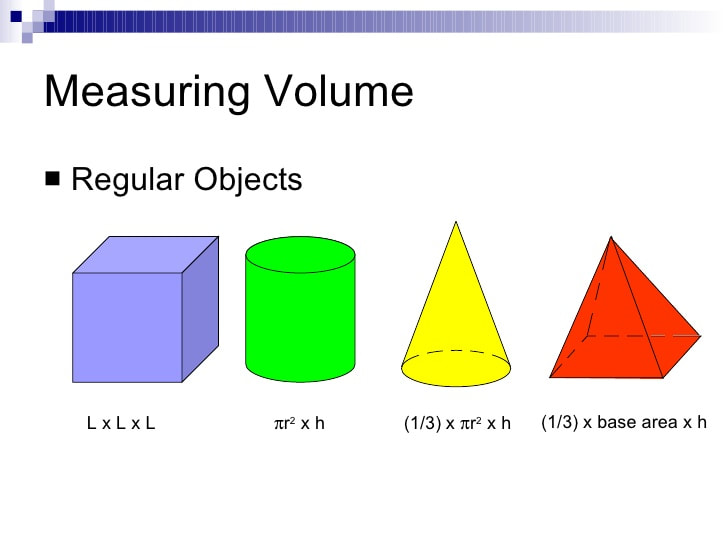
VOLUME
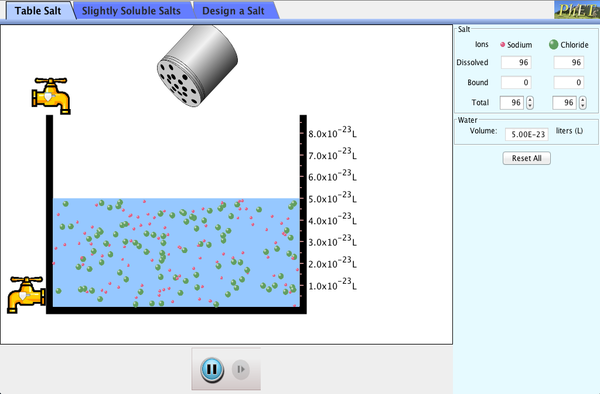
Solubility
Solubility is the property that describes how much of a substance can dissolve in another.
- High solubility = more will dissolve
- Low solubility = less will dissolve
- Insoluble/Not soluble = nothing will dissolve
Viscosity
Viscosity is the property that describes how well a substance flows.
- High viscosity = Slow flow, high resistance
- Low viscosity = fast flow, low resistance
|
|
|