-
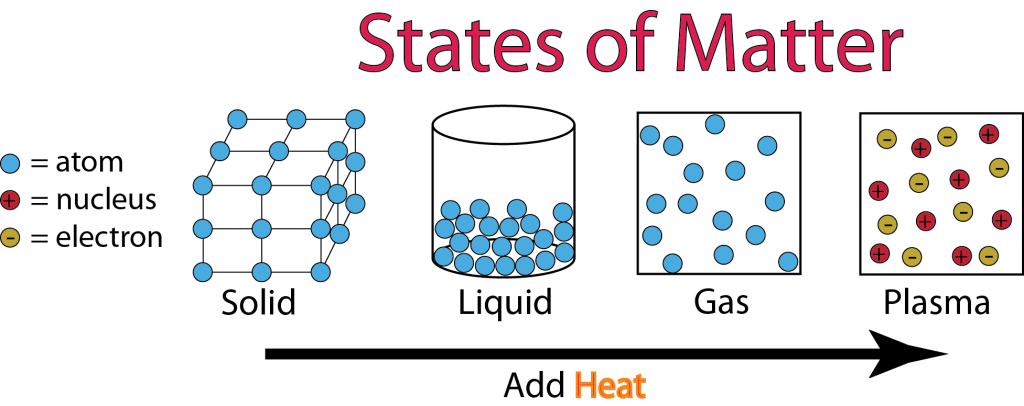
Kinetic Theory of Matter
-
Solid
-
Liquid
-
Gas
-
Plasma
<
>
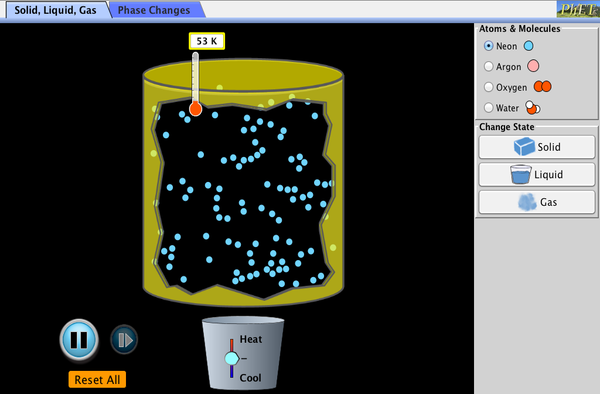
Kinetic Theory of Matter = "Atoms and molecules are always moving!"
|
|
In these two videos, you will see...
...how atoms and molecules behave in the three states of matter. ...how atoms and molecules behave when heat is added. Think about size and shape. How will the size or shape change based on the state of matter? |
|
Hold up... atoms are too small to see? How do we know all of this?
|
Think about a glass of water. Picture it sitting on the table, very still. Drop some red dye into the water... but be careful not to disturb the water too much. Now let it sit for a few minutes.
Even without stirring, the water will eventually change colors. This suggests that even though the water doesn't appear to be moving, the individual molecules are moving! What happens if you try this again using hot water and cold water? |
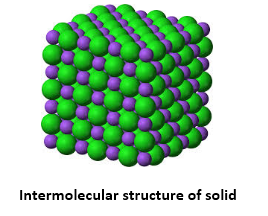
Solid:
|
Solid is a state of matter where atoms and molecules are close together. The atoms and molecules don't have enough energy to break free from each other.
Solid Size (Volume) = Definite, stays the same, won't change
Solid Shape = Definite, stays the same, won't change Intra = Inside
Intramolecule = The space between molecules |
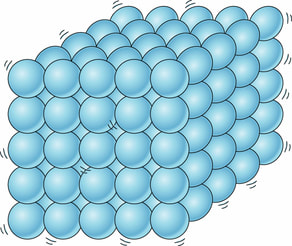
Liquid:
|
Liquid is a state of matter where atoms and molecules flow around each other. The atoms and molecules have just enough energy to slip and slide around each other, but not enough to escape or fly away.
Liquid Size (Volume) = Definite, stays the same, won't change
Liquid Shape = Indefinite, changes, takes the shape of its container |
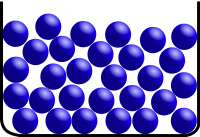
Gas:
|
Gas is a state of matter where atoms and molecules fly around with a high energy. The atoms and molecules have so much energy that they fly around wherever they can. Unless something stops them, particles in a gas will spread out forever!
Gas Size (Volume) = Indefinte, changes, expands forever
Gas Shape = Indefinite, changes, takes the shape of its container |
Bonus... PLASMA!
WARNING! DO NOT DO THIS WITHOUT AN EXPERIENCED ADULT!I accidentally melted the inside of my microwave when I tried it...
:c |
|